Due to the highly hazardous nature of Hydrochloric Acid, proper safety training is essential for persons who work with this material. Our Safe Handling Guide provides an overview of the essentials of Hydrochloric Acid safety.
About Hydrochloric Acid
Hydrochloric Acid (HCl), sometimes called Muriatic Acid, is made by the combustion of Hydrogen gas in Chlorine gas to create Hydrogen Chloride. This is then dissolved in water to create a liquid Hydrochloric Acid.
Hydrochloric Acid is a highly corrosive solution. It emits a pungent odor and strongly fumes in moist air. The solution ranges in appearance from a water-like colorlessness to slightly yellow. Concentrated Hydrochloric Acid is one of the strongest acids known. It readily attacks common metals, yielding hydrogen, which, in certain concentrations in air, may be explosive. Concentrated solutions are capable of causing severe damage to eyes and skin. The vapors are irritating to the eyes, skin and mucous membranes.
Most of the Hydrochloric Acid that is produced commercially is used for “pickling” or preparing metals for electroplating. It is also used in the production of chlorides, fertilizers and dyes, and in rubber, textile, and photography industries. Hydrochloric Acid is used in our bodies to help digest our food. It is commonly referred to as stomach acid.
Hazard Statements
- May be corrosive to metals.
- Harmful if swallowed.
- Causes severe skin burns and eye damage.
- May cause respiratory irritation.
Hydrochloric Acid Storage / Disposal
Hydrochloric Acid should be stored only in the original container, in a cool, dry, well-ventilated place. Keep the container tightly closed and locked up. Regulations for disposal may vary by location. Dispose of the contents and/or container in accordance with local/regional/national/international regulations.
Hydrochloric Acid Safety / Safe Handling
Hydrochloric Acid should be only used outdoors or in a well-ventilated area. The proper PPE (personal protective equipment) must be worn. Wash thoroughly after handling. If diluting the acid, NEVER add water to the acid, ALWAYS acid to water. If possible, add below the surface of the water.
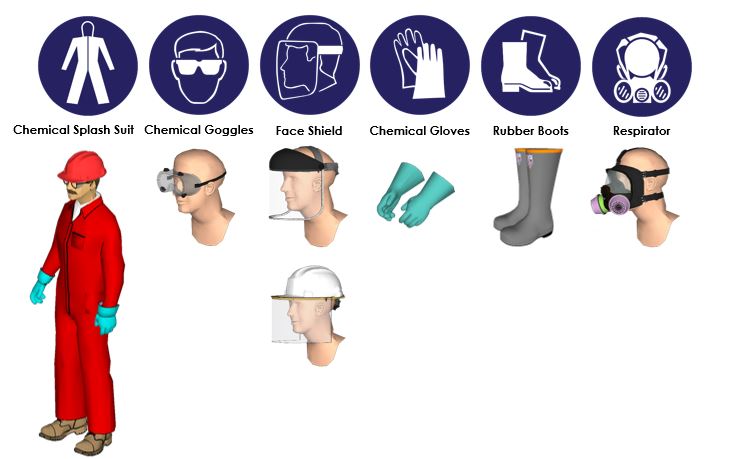
Personal Protective Equipment (PPE)
- Eyes/Face: Wear safety goggles/full face shield.
- Skin/Body: Wear a chemical-resistant suit and rubber boots.
- Hands: Wear chemical-resistant gloves.
- Respiratory: If spraying, wear an approved full-face respirator with an acid gas vapor cartridge. Do not breathe mist or vapor. A respirator is required when the concentration in the air exceeds 5 ppm.
- General: When using, do not eat, drink or smoke. Always observe good personal hygiene measures, such as washing after handling the material and before eating, drinking, and/or smoking. Routinely wash work clothing and protective equipment to remove contaminants.
Response
- If swallowed: Rinse mouth. Do NOT induce vomiting.
- If inhaled: Remove person to fresh air and keep comfortable for breathing.
- If on skin (or hair): Take off immediately all contaminated clothing. Rinse skin with water/shower.
- If in eyes: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
- Immediately call a poison center/doctor.
- Wash contaminated clothing before reuse.
- Absorb spillage to prevent material damage.
Hydrochloric Acid Spill Reporting / Neutralization
Spills greater than 5,000 lbs in a 24 hour period must be reported to the National Response Center. Spills greater than 5,000 lbs in transportation must be reported to the Department of Transportation (DOT).
| Volume of Hydrochloric Acid Spill | Amount of Sodium Bicarbonate Needed |
| 1 gallon | 5.5 lbs |
| 2 gallons | 11.0 lbs |
| 3 gallons | 16.5 lbs |
| 4 gallons | 22.0 lbs |
| 5 gallons | 27.5 lbs |
| 10 gallons | 55.0 lbs |
| 50 gallons | 275.0 lbs |
| 55 gallons | 302.5 lbs |
| 100 gallons | 550.0 lbs |
Disclaimer: This information was obtained from sources that we believe are reliable. However, the information is provided without any warranty, express or implied, regarding its correctness. The conditions or methods of handling, storage, use, and disposal of the product are beyond our control and may be beyond our knowledge. For this and other reasons, we do not assume responsibility and expressly disclaim liability for loss, damage or expense arising out of or in any way connected with the handling, storage, use or disposal of the product.
