Bleach, also known as Sodium Hypochlorite, is a widely known and used chemical product, both in the home and in industrial applications. It is known for its ability to disinfect, remove mold and mildew, and remove many types of stains. It is a wonderfully useful product when used correctly. If you’re thinking about cleaning with bleach, here are some things you need to know.

Types of Bleach
The first thing you need to know about bleach is that not all bleach is created equal. There are two different types of bleach, chlorine bleach and non-chlorine bleach. Additionally, there are different strengths of bleach. The percent of Sodium Hypochlorite in the product you are using has a huge effect on the overall performance of the product.
Chlorine Bleach: This is the product most people think of when they think of ‘bleach’. The chlorine molecule is the workhorse of chlorine bleach. Besides chlorine, it also contains water, some Sodium Hydroxide, and other things there to help keep the chlorine molecule in solution.
- Household Bleach: Bleach designed for use in the home is sold in retail and grocery stores. It is typically labeled somewhere in the range of 4-8% Sodium Hypochlorite, and is the only type of bleach appropriate for the average consumer to use at home. Due to the longer time (weeks? months?) from production to use by the customer, it is highly possible that bleach purchased in grocery stores is not necessarily as strong as is claimed on the label (see next section on bleach degradation).
- Industrial Strength Bleach: This is the only type of bleach supplied by CORECHEM Inc. As it is designed for use in industrial applications, it is much stronger. Our Sodium Hypochlorite is labeled at 12.5%, but in actuality often is a higher percentage when purchased (approx. 13.5% Sodium Hypochlorite weight percent- or trade percent 15.5%). Industrial-strength bleach is used in various industrial applications including water/wastewater treatment, disinfection and cleaning, pressure washing, etc. Due to the higher strength of the product, it should only be used by persons who understand the material and its hazards.
Non-Chlorine Bleach: This is a blend of several other chemicals. It does not contain the chlorine molecule, and is not produced from the chlor-alkali process. It may contain some combination of sodium percarbonate, sodium perborate, borax, and/or soda ash. This may be specified for use for certain applications, such as the washing of color-fast textiles/laundry. It is available in retail stores for use in home consumption and used in the textiles industry.
Does Bleach Degrade?
Chlorine bleach starts to degrade the moment it is produced. Chlorine bleach is produced via the chlor-alkali process at an elevated temperature. It is not a stable solution, meaning that it cannot exist in the same state indefinitely. The chlorine molecule is a diatomic gas molecule. This means that it is in its happiest state when it is joined up with itself. Exposure to heat and light gives it the inertia or energy needed for the chlorine molecules to escape the solution and join with itself as a gas, causing bleach degradation. The higher the temperature, the faster bleach will degrade. Even 10 degrees can make a huge difference in maintaining your bleach’s effectiveness. See the chlorine degradation chart below for an illustration of this.
Estimated Decomposition of 12.5 Wt. % Sodium Hypochlorite Solution
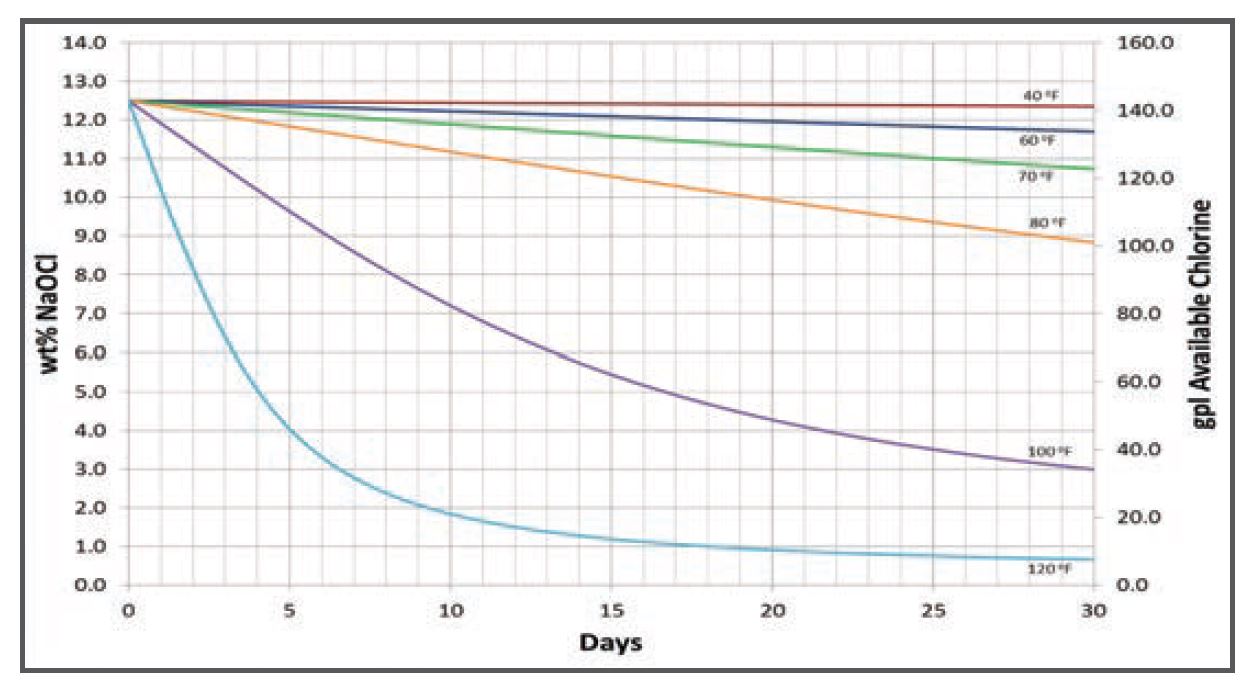
Does this mean that bleach would be best stored cold or chilled? No, not necessarily. If the temperature gets too low, salt crystals will precipitate out of the solution.
For the most effective bleach, it is important to source the freshest bleach possible. This is why, for the industrial buyer, it is recommended to buy bleach from a chemical distributor who sells fresh bleach and has strict quality controls in place (such as CORECHEM), rather than buying it from the store.
After bleach has degraded to a lower concentration, the rate of degradation slows. It takes more time for chlorine molecules to join up with other chlorine molecules and flee the solution when it is weaker.
Non-chlorine bleach is stable and does not degrade.
How To Clean With Bleach
Bleach is very effective when used for disinfection, mold or mildew removal, whitening whites, and other appropriate applications. However, it does not actually remove dirt and soil on its own, and it is not a surfactant. An area that will be cleaned with bleach should be washed and rinsed first to remove any dirt and grime before using the bleach solution.
To clean with bleach, create a solution of the desired concentration. Different concentrations are recommended for different applications. A ‘standard’ formula for home use is ½ cup bleach to 1 gallon of water. This may vary depending on what you are cleaning. Outdoor trash cans may need a stronger solution; a much weaker solution may be used for items like kitchen cutting boards.
A bleach solution can be applied by either soaking a rag and wiping (using plenty of water, enough to keep the area wet for a minute or so to properly disinfect it), or by soaking items directly in the solution. If using a solution, let sit for 5 – 10 minutes. Bleach begins working immediately on contact, but needs some time to do its job. After soaking or wiping, rinse well and let dry.
Do not let bleach come in contact with other chemicals or products unless you know what you are doing as hazardous gases can be created. After the area being cleaned is fully rinsed and dry, you can safely use other products on it without waiting.
Professional pressure washers often use bleach mixed with a surfactant such as Dawn dish soap to help the product stick to the houses and areas they are cleaning. This is fine, but note that the soap and bleach are incompatible being together in solution long term. If using this way, mix the solution fresh at the job each time.
How Bleach Works
Chlorine Bleach: There are several things included in a Sodium Hypochlorite solution. The chlorine molecule (originally a gas), water, small amounts Caustic Soda left over from the manufacturing process, and other things there to try to keep the chlorine molecule happy in a solution for an extended period of time. The elemental chlorine molecule is the workhorse of chlorine bleach. It works as an oxidizing agent. Oxidizing agents react with surfaces and break chemical bonds which help to kill germs and whiten pigments.
When light hits a color and we see it, this is because the fabric is absorbing the other colors while reflecting that color back into your eye. Chlorine whitens and removes stains by removing electrons from the dye and/or textile molecules. By taking a few electrons off of a molecule, it will strip it of its ability to change light.
Non-Chlorine Bleach: Non-chlorine bleach is not an oxidizing agent, but a reducing agent. These add electrons back to certain molecules.
What Can I Clean With Bleach?
Bleach can be used to clean in a wide variety of applications. It is important to note that the proper concentration for the application is crucial. Bleach used at too strong a concentration for the application is harsh and can ruin things like textiles, wood, etc.
Some areas bleach may be useful around the home would include: in pools, on stainless steel, on grout, in your dishwasher, in your washing machine, on quartz countertops, on white clothes/sheets/towels, in the shower, on dishes, on trash cans, in white kitchen sinks, on patio furniture, etc. It can also be used to kill drain flies, yellow jackets, and will destroy the pheromone scent of bugs such as bees, ants, and roaches.
It can kill weeds and grass at stronger concentrations. If you are cleaning outdoors and don’t want it to kill the grass, dilute with plenty of water.
Professional pressure washers use it on concrete, home exteriors, and other appropriate outdoor areas.
Bleach may eat coatings off of hardwood floors if they are not robust enough, or if there are any imperfections in the surface, so we don’t recommend this use.
Wear Protective Equipment When Cleaning With Bleach
Bleach is a very useful product when used correctly, but can result in grave harm or injury if handled incorrectly. Regardless of strength, appropriate precautions need to be taken when using it. Proper personal protective equipment such as eye protection and protective gloves should be worn. Always use with good ventilation. Keep kids and pets away while using bleach. For full safety information, consult the Safety Data Sheet.
For more information on sales of industrial-strength Bleach, see Sodium Hypochlorite 12.5%.
