Sulfuric Acid is a highly corrosive liquid mineral acid. Proper safety training, as well as a strong awareness of the inherent dangers of the chemical, are essential to those who work with it. Our Safe Handling Guide provides an overview of the essentials of Sulfuric Acid safety.
About Sulfuric Acid
Sulfuric Acid is a very strong inorganic acid. It is heavy, oily, has a pH of less than 1, and has an acrid odor. Pure Sulfuric Acid looks just like water and has no fumes. Technical grades may be off-white or turbid in appearance. When it comes in contact with water, heat is generated. Sulfuric Acid is corrosive and has the ability to cause severe chemical burns.
There are several ways Sulfuric Acid may be produced. Some methods include the contact process, lead chamber process, wet sulfuric acid process, and spent acid regeneration process. It is often produced by burning elemental sulfur.
Sulfuric Acid has uses in many different applications and industries. It is considered essential in industries such as fertilizers, textiles, pulp and paper, pigments and dyes, steel, metals, car batteries, chemical manufacturing, as well as many others. It is such an important commodity chemical that more Sulfuric Acid is produced in America (the top chemical producer in the world) than any other chemical.
Hazard Statements
- May be corrosive to metals.
- Causes severe skin burns and eye damage.
- Causes serious eye damage.
- May cause respiratory irritation.
Sulfuric Acid Storage / Disposal
Sulfuric Acid should be stored in containers specially designed for the product and strength. Store the product away from incompatible materials, locked up, and in a well-ventilated place. Keep away from heat, sparks, and open flame. This material and its container must be disposed of as hazardous waste. Collect and reclaim or dispose in sealed containers at a licensed waste disposal site. Do not allow this material to drain into sewers/water supplies. Do not contaminate ponds, waterways, or ditches with chemical or used container. Regulations for disposal may vary by location. Dispose of contents/container in accordance with local/regional/national/international regulations.
Sulfuric Acid Safety / Safe Handling
Sulfuric Acid is a strong acid that can cause severe chemical and thermal burns to the skin and eyes. If mist or vapor is inhaled, it can cause lung injury, cough, difficulty breathing, and shortness of breath. For this reason, it is very important that appropriate personal protective equipment (PPE) is worn at all times while handling this material, so that material does not come in contact with skin, eyes, clothing, etc; and that adequate ventilation is used. In addition, good industrial hygiene practices should be observed at all times.
When diluting Sulfuric Acid, use caution, as it reacts with water and generates heat. Always add the acid to the water slowly while mixing to minimize heat generation.
When handling, transferring or diluting Sulfuric Acid, make sure to use equipment that is compatible with your application. Dilute solutions of Sulfuric Acid are very corrosive to most metals and special materials of construction must be used. If diluting, the equipment used must be able to tolerate the heat generated as well as the corrosivity of the final solution.
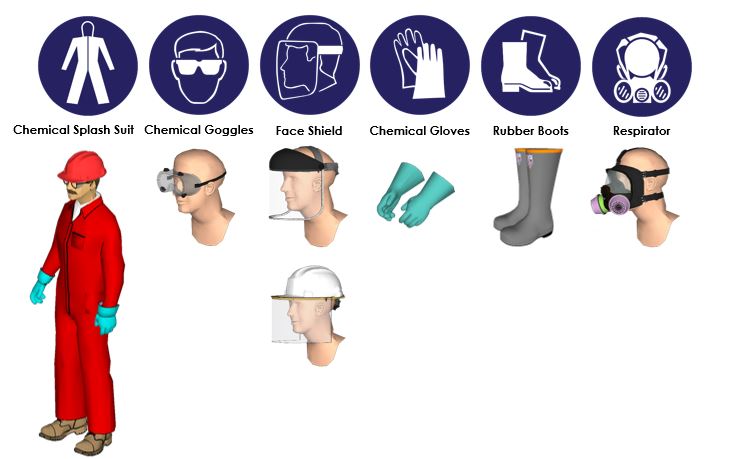
Personal Protective Equipment (PPE)
The level of protection required may vary depending on the level of exposure the employee is going to experience. Consider the PPE recommendations below for tasks such as: Entering the area where acid is handled, reading gauges, inspecting equipment, operating valves, switching on pumps, making line breaks into a Sulfuric Acid system, and connecting/disconnecting hoses.
- Eyes/Face: Wear close-fitting chemical splash goggles that do not allow entry of liquids. They should have adequate ventilation to keep them from fogging. Use a face shield or splash hood to protect your face from possible splashing. Wear a hard hat.
- Skin/Body: Wear a one or two-piece acid suit that is rated for use with Sulfuric Acid, and acid-resistant boots. Wear pant legs over boots.
- Hands: Wear acid-resistant gauntlet-length gloves with sleeves over gloves.
- Respiratory: Wear a respirator if exposure is expected to exceed regulatory limits.
- General: Always observe good personal hygiene measures, such as washing after handling the material and before eating, drinking, and/or smoking. Do not eat, drink, or smoke while handling. Routinely wash work clothing and protective equipment to remove contaminants. PPE should not be worn or carried beyond the operating area. Wash before reuse. Discard contaminated footwear that cannot be cleaned.
In the event of a large leak or spill, additional protection may be needed. On no occasion should employees enter an area where they may be sprayed with a stream of acid or step into acid puddles.
Response
First aid for any type of exposure must be started immediately.
- If swallowed: Rinse mouth. Do NOT induce vomiting. Immediately give the patient large quantities of water. Never give anything by mouth to an unconscious person.
- If inhaled: Move to fresh air and keep at rest in a position comfortable for breathing. Give oxygen if breathing is difficult. Artificial respiration may be applied if necessary.
- If on skin (or hair): Take off immediately all contaminated clothing and rinse skin with plenty of water (preferably cold water) for at least 15 minutes. Since Sulfuric Acid generates heat when in contact with water, severe burning may occur during the water clean-up process. Do not scrape or wipe acid off of skin. If medical treatment must be delayed, apply ice water compresses or immerse the affected area in ice water.
- If in eyes: Rinse cautiously with water for at least 15 minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
- Immediately call a poison center/doctor.
- Wash contaminated clothing before reuse.
Sulfuric Acid Spill Reporting / Neutralization
In the event of a Sulfuric Acid spill, immediately zone off the area and close valves. Keep people away from and upwind of the spill area; evacuate employees to safe areas. Before attempting clean-up, make sure PPE is appropriate and sufficient. Remove all possible sources of ignition. Contain spill to keep out of sewers. If available, apply a fine water mist or mid-expansion foam to the contaminated area. If not available, sand, ashes, or gravel may be used. Neutralize the acid with some alkaline material such as lime, sodium bicarbonate, or soda ash. Consult with the applicable government authorities about spill reporting requirements.
Disclaimer: This information was obtained from sources that we believe are reliable. However, the information is provided without any warranty, express or implied, regarding its correctness. The conditions or methods of handling, storage, use, and disposal of the product are beyond our control and may be beyond our knowledge. For this and other reasons, we do not assume responsibility and expressly disclaim liability for loss, damage or expense arising out of or in any way connected with the handling, storage, use or disposal of the product.
