Sodium Hydroxide is a highly caustic base and alkali. Proper safety training, as well as a strong awareness of the inherent dangers of the chemical, are essential to those who work with it. Our Safe Handling Guide provides an overview of the essentials of Sodium Hydroxide safety.
About Sodium Hydroxide
Liquid Sodium Hydroxide, sometimes called Caustic Soda, is most commonly made via the electrolysis of brine (Sodium Chloride). Brine enters either a membrane or diaphragm electrolytic cell, and three products are produced: Caustic, Chlorine, and Hydrogen. This process is known as the Chlor-alkali process.
Sodium Hydroxide Solution is highly corrosive, and a very strong base. (See: What is pH?) It may be clear or slightly cloudy, depending on the grade; see Sodium Hydroxide: Membrane vs Diaphragm Grade. It is often transported and handled hot. If Caustic comes in contact with water, an exothermic reaction will occur. Sodium Hydroxide is known for its ability to cause severe chemical burns. Contact with the skin will cause severe burns, and the inhalation of vapors is extremely irritating.
Sodium Hydroxide has uses in very many different applications and industries. Commonly, it is used in the pulp and paper industry, for water treatment, for pH regulation, and in food preparation. Sodium Hydroxide is so diverse it may be used for anything from giving pretzels their crispy crust, to the unsavory digestion of roadkill!
Hazard Statements
- May be corrosive to metals.
- Harmful if swallowed.
- Causes severe skin burns and eye damage.
- Causes serious eye damage.
Sodium Hydroxide Storage / Disposal
Sodium Hydroxide should be stored in the original container and locked up. The container should be a corrosive-resistant container with a resistant inner liner. Keep the container tightly closed and store it in a cool, dry, well-ventilated place. Regulations for disposal may vary by location. Dispose of the contents and/or container to an appropriate treatment and disposal facility in accordance with local/regional/national/international regulations, and product characteristics at the time of disposal.
Sodium Hydroxide Safety / Safe Handling
Use caution when diluting Sodium Hydroxide. To minimize heat generation, DO NOT add water to Caustic; ALWAYS add Caustic to water while stirring. When handling Sodium Hydroxide, it is important that proper PPE (personal protective equipment) is worn. This product must not come in contact with eyes, skin, or clothing. Use only with adequate ventilation; do not inhale mist or vapors, and do not taste or swallow.
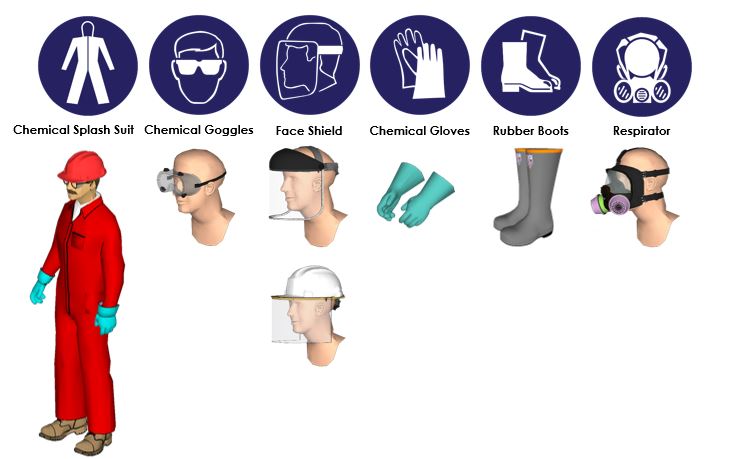
Personal Protective Equipment (PPE)
- Eyes/Face: Wear close-fitting chemical splash goggles that do not allow entry of liquids. They should have adequate ventilation to keep them from fogging. Use a face shield to protect your face from possible splashing.
- Skin/Body: Wear a chemical-resistant suit and rubber boots. Do not wear leather boots as Caustic disintegrates leather. Do NOT tuck pant legs into boots.
- Hands: Wear chemical-resistant rubber or rubber/plastic-coated gloves that come well above the wrist.
- Respiratory: Wear a respirator if exposure is expected to exceed regulatory limits.
- General: When handling, do not eat, drink or smoke. Always observe good personal hygiene measures, such as washing after handling the material and before eating, drinking, and/or smoking. Routinely wash work clothing and protective equipment to remove contaminants.
Response
- If swallowed: Rinse mouth and drink plenty of water. Do NOT induce vomiting. If vomiting occurs, keep head low so that stomach content doesn’t get into the lungs.
- If inhaled: Move to fresh air. If breathing is difficult, give oxygen. If breathing stops, provide artificial respiration. Do not use mouth-to-mouth.
- If on skin (or hair): Take off immediately all contaminated clothing and wash off with plenty of water for 15-20 minutes.
- If in eyes: Immediately flush with water for at least 15 minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
- IMMEDIATELY call a poison center/doctor.
- Wash contaminated clothing separately before reuse.
Sodium Hydroxide Spill Reporting / Neutralization
Contain and collect spill to be removed to a chemical waste area. Neutralize the residue with a dilute or weak acid. Flush spill area with water. Notify the applicable government authority if the spill is reportable or if it could harm the environment.
Disclaimer: This information was obtained from sources that we believe are reliable. However, the information is provided without any warranty, express or implied, regarding its correctness. The conditions or methods of handling, storage, use, and disposal of the product are beyond our control and may be beyond our knowledge. For this and other reasons, we do not assume responsibility and expressly disclaim liability for loss, damage or expense arising out of or in any way connected with the handling, storage, use or disposal of the product.
