Liquid Sodium Hydroxide, or Caustic Soda, is made via the electrolysis of brine in a process known as the chlor-alkali process. During electrolysis, an electrical current is used to cause an otherwise non-spontaneous chemical reaction; in this case, the decomposition of salt.
What does this look like? Brine passes into an electrolytic cell. The cell is divided into two compartments by either a membrane or a diaphragm. A positively charged anode is in one compartment, and a negatively charged cathode is in the other. The salt water enters the anode compartment, then flows into the cathode compartment. Multiple chemicals are created in the process; the barrier separates the chemicals that are being created. The Sodium Hydroxide that is produced will be either ‘Membrane Grade’ or ‘Diaphragm Grade’, depending on the type of cell that was used to create it.
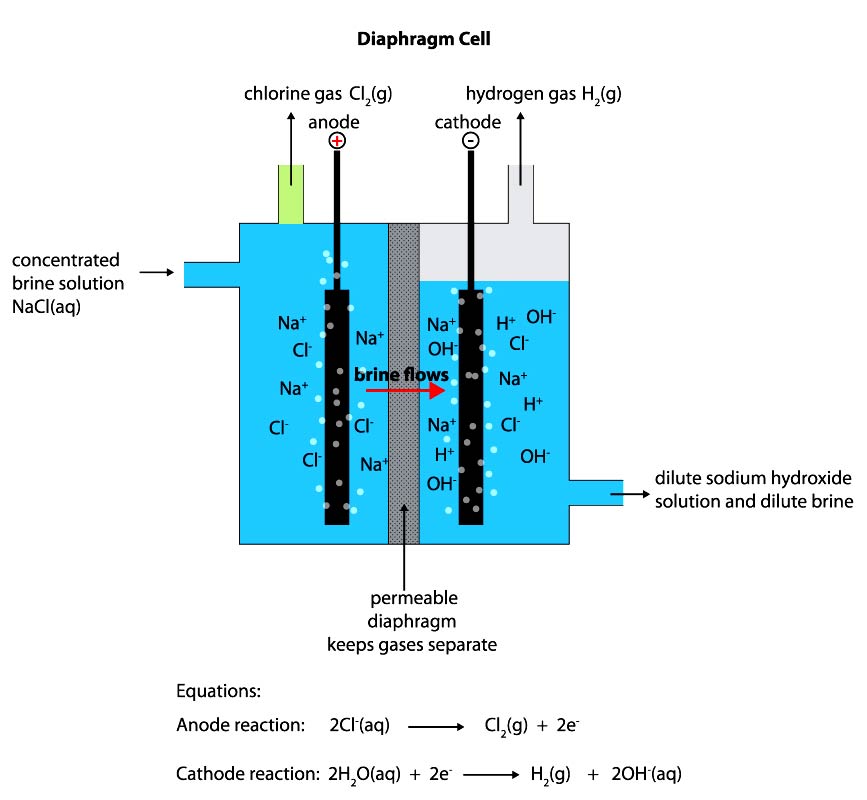
Diaphragm Grade Caustic Soda: Diaphragm cells have been in use for over 100 years. The diaphragm barrier is permeable and commonly made of asbestos fibers. Brine flows into the anode compartment, where chlorine is created; then into the cathode compartment, where a dilute sodium hydroxide is created. The resulting Sodium Hydroxide product is a weak concentration (approx. 12%), and contains a significant amount of salt. Evaporation crystallizes out much of the salt and brings the solution to a higher Sodium Hydroxide concentration; however, the final product is still known for it’s higher salt content. Due to higher salt content, Diaphragm Grade Caustic Soda may appear more cloudy. As salt is corrosive to machinery and equipment, Diaphragm Grade Caustic Soda is often less desirable for use by certain industries, i.e. pulp and paper. The use of asbestos in the diaphragm also make this a less environmentally friendly process.
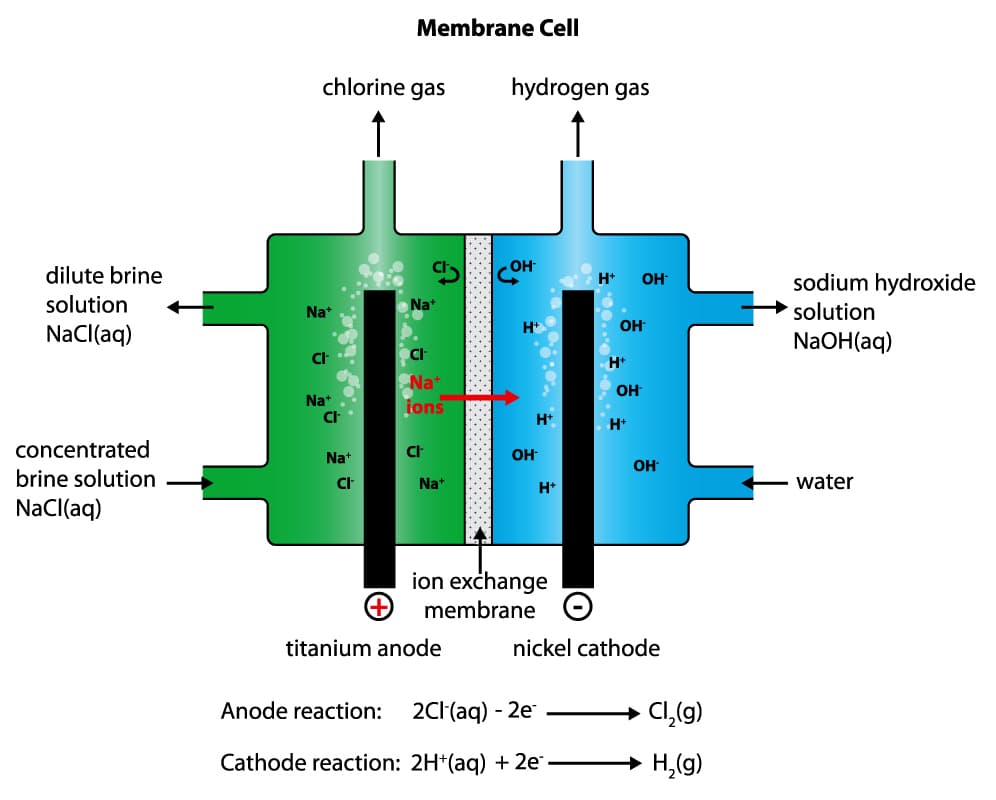
Membrane Grade Caustic Soda: Membrane cells have only been developed in the past 60 years. Similar to the diaphragm process, chlorine is created in the first compartment of the cell at the anode, and caustic is created in the second compartment at the cathode. The membrane barrier that divides the cell is made from a polymer. Positively charged sodium ions are able to pass through it, but the negatively charged chloride ions cannot. The resulting Sodium Hydroxide that is produced is a much higher concentration (approx. 33%), and contains much less salt than caustic produced in a diaphragm cell. This material is then evaporated to increase the concentration to 50%. Membrane Grade Caustic Soda is usually clearer in appearance and contains an overall lower level of impurities compared to Diaphragm Grade material. Due to increased energy efficiency, greater environmental friendliness, and higher purity of product, the membrane cell is the more commonly used process today.
